 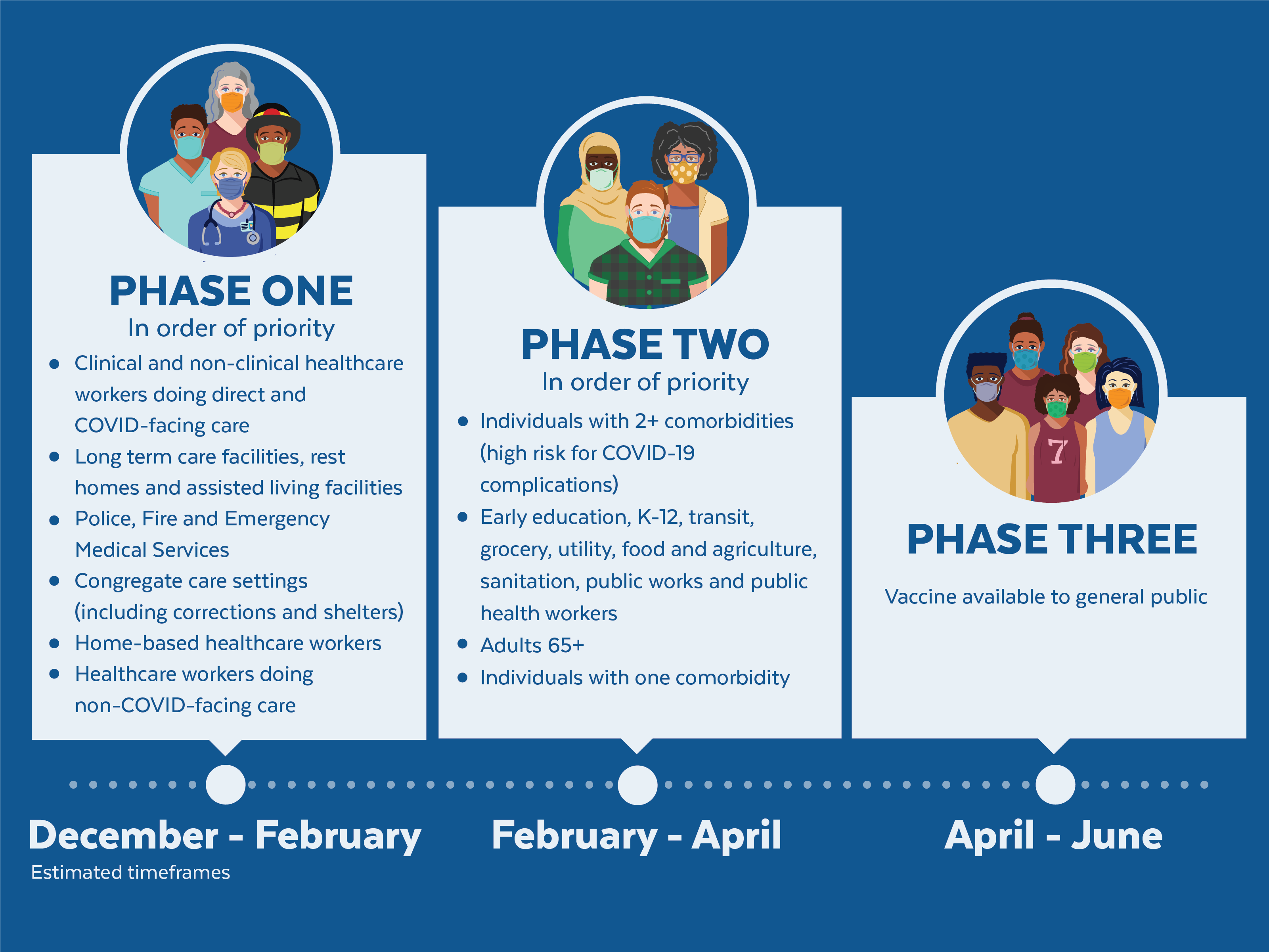
The Centers for Disease Control and Prevention (CDC) required all states and territories to turn in an interim. The vaccine is available under emergency use authorization for teens and children age 6 months and older. Moderna, Inc., mRNA-1273: On January 31, 2022, the FDA approved this NIH-funded COVID-19 vaccine called Spikevax for people age 18 and older. Would we shut down again? What will the United States do the next time a deadly virus comes knocking on the door?įor the latest news, sign up for our free newsletter. District of Columbia COVID-19 Vaccination Plan. Immunization against Pertussis (whooping cough), COVID-19 and flu are an essential component. View the infographic to see what is in the Pfizer COVID-19 vaccine. 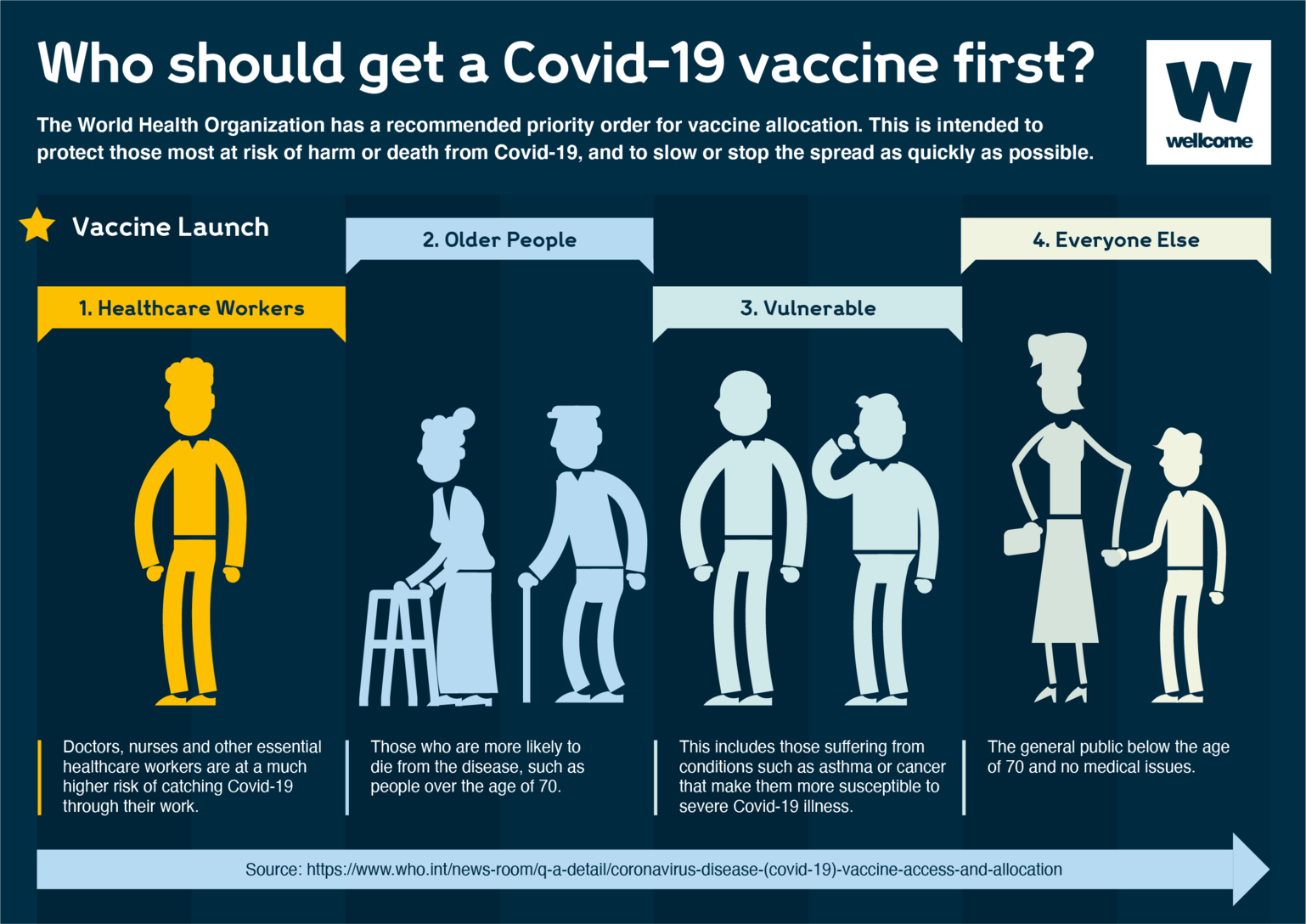
Here’s what you need to know about Arcturus. The latest omicron offshoot is particularly prevalent in India. Most individuals with certain kinds of immunocompromise who have received a bivalent COVID-19 vaccine may receive a single additional dose of a bivalent COVID-19 vaccine at least 2 months. 1.16, has been designated as a “variant under monitoring” by the World Health Organization. New covid variant: A new coronavirus subvariant, XBB. Here’s who should get the second covid booster and when. The latest on coronavirus boosters: The FDA cleared the way for people who are at least 65 or immune-compromised to receive a second updated booster shot for the coronavirus. 
Tracking covid cases, deaths: Covid-19 was the fourth leading cause of death in the United States last year with covid deaths dropping 47 percent between 20. Here’s what the end of the covid public health emergency means for you. End of the public health emergency: The Biden administration ended the public health emergency for the coronavirus pandemic on May 11, just days after WHO said it would no longer classify the coronavirus pandemic as a public health emergency. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
